Bisphosphonates are a type of drug/medication which blocks the loss of bone density to treat osteoporosis-related ailments. They are most frequently prescribed for the treatment of osteoporosis. Bisphosphonates have two phosphonate groups. Evidence demonstrates that they reduce the probability of fractures in post-menopausal women with osteoporosis.
Bone tissue undergoes continuous remodeling that is stored to provide equilibrium, or homeostasis, through osteoblasts generating bone and osteoclasts ruining bone. Bisphosphonates inhibit bone digestion by encouraging osteoclasts to undergo apoptosis or cell death.
The uses of bisphosphonates include the prevention and treatment of osteoporosis, Paget’s disease of bone, bone metastasis (with or without hypercalcaemia), multiple myeloma, primary hyperparathyroidism, osteogenesis imperfecta, fibrous dysplasia, and other conditions which exhibit bone fragility. The purpose of the following article is to discuss the mechanism of action and role in the clinical practice of bisphosphonates.
Table of Contents
Abstract
Bisphosphonates are primary agents in the current pharmacological arsenal against osteoclast-mediated bone loss due to osteoporosis, Paget disease of bone, malignancies metastatic to bone, multiple myeloma, and hypercalcemia of malignancy. In addition to currently approved uses, bisphosphonates are commonly prescribed for
Introduction
Since their introduction to clinical practice more than 3 decades ago, bisphosphonates have been increasingly used for an array of skeletal disorders. Bisphosphonates are now used to treat such varied conditions as heritable skeletal disorders in children, postmenopausal and glucocorticoid-induced osteoporosis (GIO), and bone metastases in patients with malignancies. Bisphosphonates can offer substantial clinical benefit in conditions in which an imbalance between osteoblast-mediated bone formation and osteoclast-mediated bone resorption underlies disease pathology; however, the more recently recognized association of bisphosphonate use with pathologic conditions, including low bone turnover states with resultant pathologic fractures, osteonecrosis of the jaw (ONJ), and an increased incidence of atrial fibrillation, has brought increased scrutiny to the current broad use of bisphosphonate therapy.
PubMed literature from January 1, 1998, to May 1, 2008, was reviewed using bisphosphonate and clinical practice as search terms. Additional articles not obtained in the primary search were identified by assessment of literature referenced in the reviewed articles. We present data on the development of bisphosphonates as therapeutic agents, the proposed mechanisms by which these agents exert their effects, and the current roles for bisphosphonate therapy in clinical practice. Additionally, we address some areas of concern for clinicians and draw attention to some currently unresolved issues associated with bisphosphonate use.
Chemical Structure as Basis for Clinical Activity
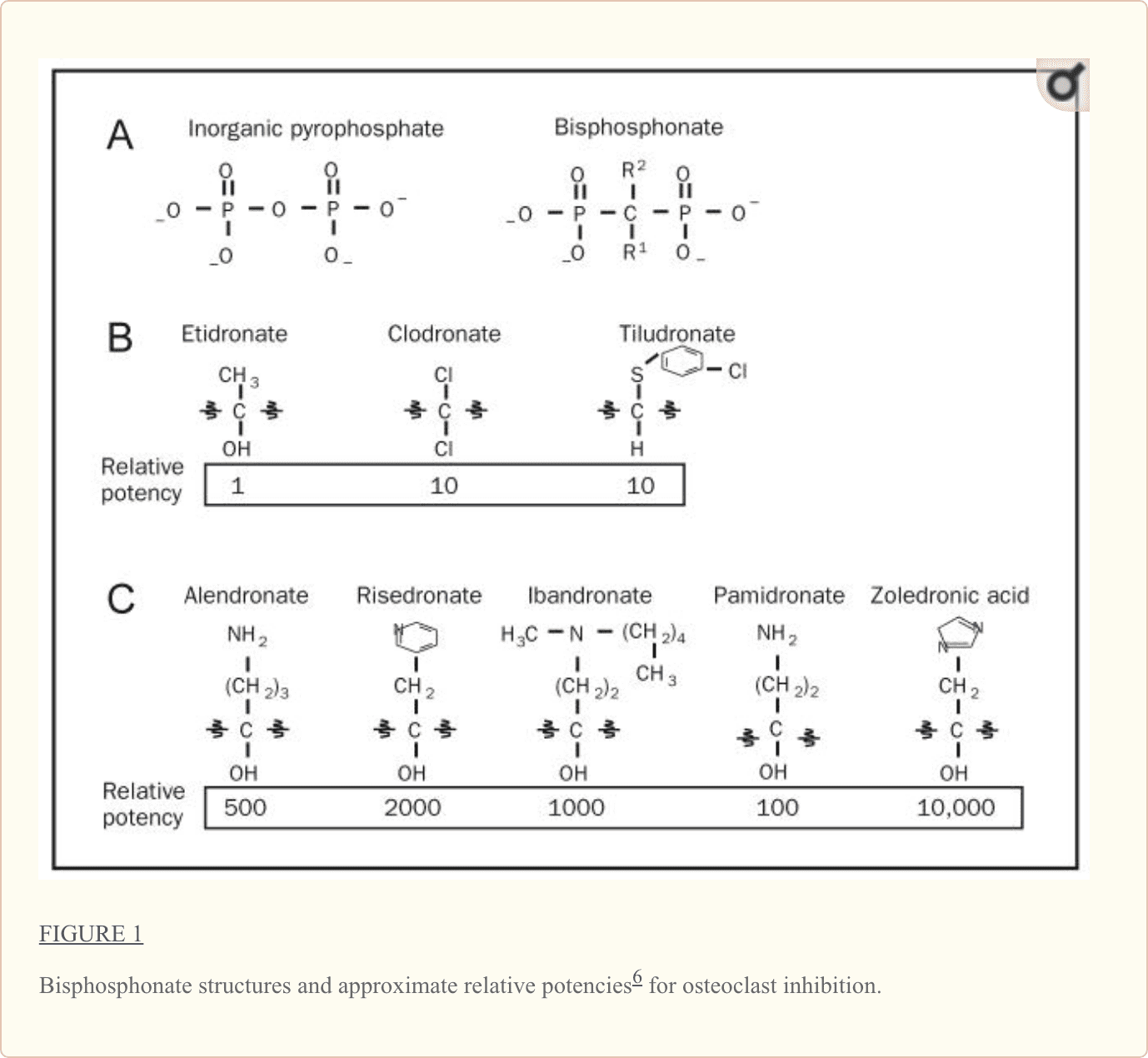
Structurally, bisphosphonates are chemically stable derivatives of inorganic pyrophosphate (PPi), a naturally occurring compound in which 2 phosphate groups are linked by esterification (Figure 1, A). Within humans, PPi is released as a by-product of many of the body’s synthetic reactions; thus, it can be readily detected in many tissues, including blood and urine.1 Pioneering studies from the 1960s demonstrated that
Like their natural analogue PPi, bisphosphonates have a very high affinity for bone mineral because they bind to hydroxyapatite crystals. Accordingly, bisphosphonate skeletal retention depends on availability of hydroxyapatite binding sites. Bisphosphonates are preferentially incorporated into sites of active bone remodeling, as commonly occurs in conditions characterized by accelerated skeletal turnover. Bisphosphonate not retained in the skeleton is rapidly cleared from the circulation by renal excretion. In addition to their ability to inhibit calcification, bisphosphonates inhibit hydroxyapatite breakdown, thereby effectively suppressing bone resorption.3 This fundamental property of bisphosphonates has led to their utility as clinical agents. More recently, it has been suggested that bisphosphonates also function to limit both osteoblast and osteocyte apoptosis.4,5 The relative importance of this function for bisphosphonate activity is currently unclear.
Modification of the chemical structure of bisphosphonates has widened the differences between the effective bisphosphonate concentrations needed for antiresorptive activity relative to those that inhibit bone matrix mineralization, making the circulating concentrations of all bisphosphonates currently used in clinical practice active essentially only for the inhibition of skeletal resorption.1 As shown in Figure 1, A, the core structure of bisphosphonates differs only slightly from PPi in that bisphosphonates contain a central nonhydrolyzable carbon; the phosphate groups flanking this central carbon are maintained. As detailed in Figure 1, B, and distinct from PPi, nearly all bisphosphonates in current clinical use also have a hydroxyl group attached to the central carbon (termed the R1 position). The flanking phosphate groups provide bisphosphonates with a strong affinity for hydroxyapatite crystals in bone (and are also seen in PPi), whereas the hydroxyl motif further increases a bisphosphonate’s ability to bind calcium. Collectively, the phosphate and hydroxyl groups create a tertiary rather than a binary interaction between the bisphosphonate and the bone matrix, giving bisphosphonates their remarkable specificity for bone.1
Although the phosphate and hydroxyl groups are essential for bisphosphonate affinity for bone matrix, the final structural moiety (in the R2 position) bound to the central carbon is the primary determinant of a bisphosphonate’s potency for inhibition of bone resorption. The presence of a nitrogen or amino group increases the bisphosphonate’s antiresorptive potency by 10 to 10,000 relative to early non–nitrogen-containing bisphosphonates, such as etidronate.1,6 Recent studies (described subsequently) delineate the molecular mechanism by which nitrogen-containing bisphosphonates inhibit osteoclast activity.
A critical pharmacological feature of all bisphosphonates is their extremely high affinity for, and consequent deposition into, bone relative to other tissues. This high affinity for bone mineral allows bisphosphonates to achieve a high local concentration throughout the entire skeleton. Accordingly, bisphosphonates have become the primary therapy for skeletal disorders characterized by excessive or imbalanced skeletal remodeling, in which osteoclast and osteoblast activities are not tightly coupled, leading to excessive osteoclast-mediated bone resorption.
Early non–nitrogen-containing bisphosphonates (etidronate, clodronate, and tiludronate) (Figure 1, B) are considered first-generation bisphosphonates. Because of their close structural similarity to PPi, non–nitrogen-containing bisphosphonates become incorporated into molecules of newly formed adenosine triphosphate (ATP) by the class II aminoacyl–transfer RNA synthetases after osteoclast-mediated uptake from the bone mineral surface.1 Intracellular accumulation of these nonhydrolyzable ATP analogues is believed to be cytotoxic to osteoclasts because they inhibit multiple ATP-dependent cellular processes, leading to osteoclast apoptosis.
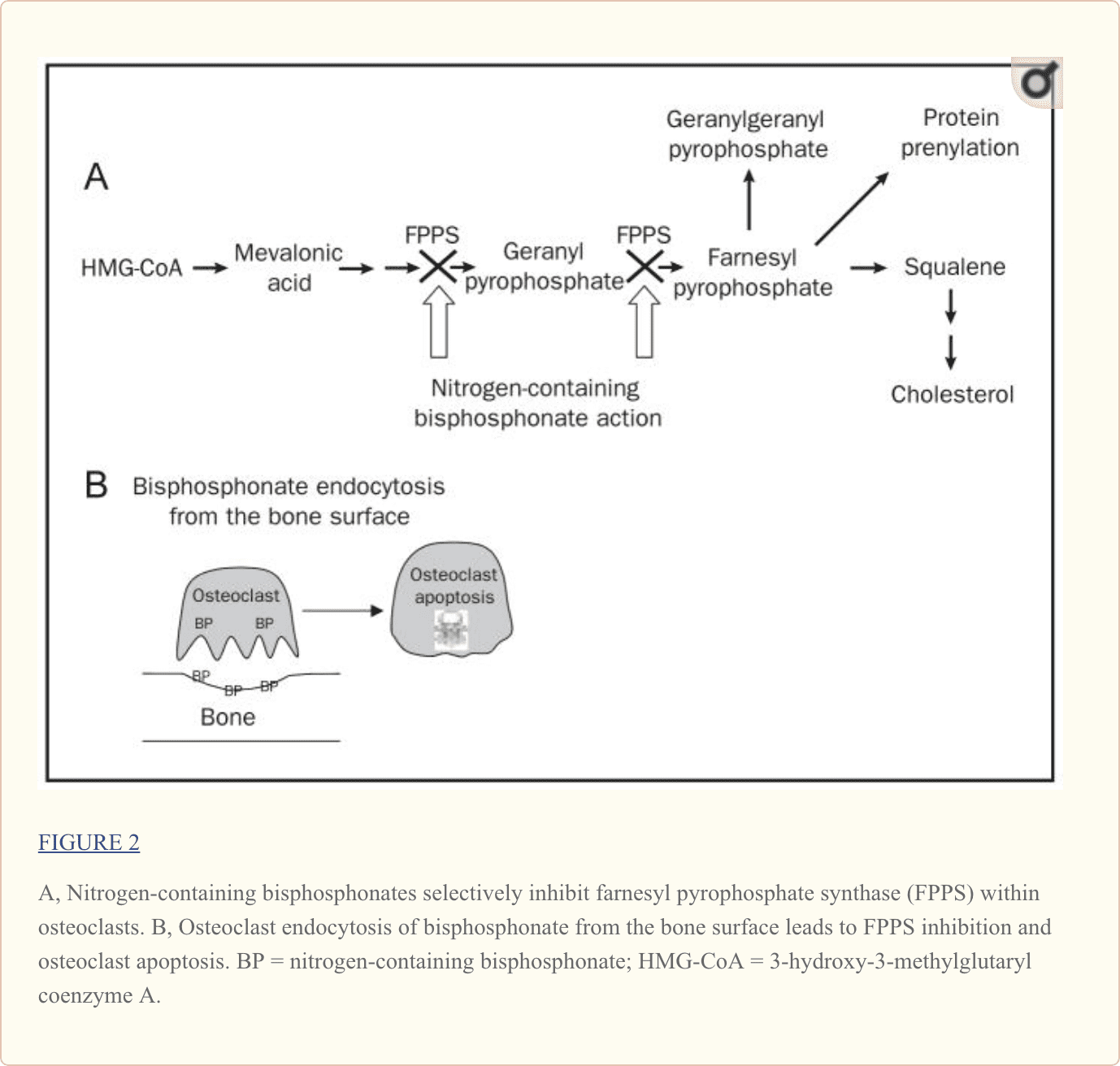
Unlike early bisphosphonates, second- and third-generation bisphosphonates (alendronate, risedronate, ibandronate, pamidronate, and zoledronic acid) have nitrogen-containing R2 side chains (Figure 1, C). The mechanism by which nitrogen-containing bisphosphonates promote osteoclast apoptosis is distinct from that of the non–nitrogen-containing bisphosphonates. As elegantly illustrated in recent studies, nitrogen-containing bisphosphonates bind to and inhibit the activity of farnesyl pyrophosphate synthase, a key regulatory enzyme in the mevalonic acid pathway critical to the production of cholesterol, other sterols, and isoprenoid lipids6,7 (Figure 2, A). the analog is likely a direct function of the ability of bisphosphonates to selectively adhere to and be retained within bone before endocytosis within osteoclasts during osteoclast-mediated bone mineral dissolution and matrix digestion (Figure 2, B). Given the fact that nearly all patients now receive treatment with the more potent nitrogen-containing bisphosphonates rather than the earlier non–nitrogen-containing bisphosphonates, the remainder of this review focuses on this more recent class of bisphosphonates.
Additional Clinical Features
Although bisphosphonate-mediated induction of osteoclast apoptosis cannot be measured directly within the clinical setting, a temporal reduction in biochemical markers of bone resorption (namely amino- and carboxyl-terminal breakdown products of type 1 collagen in serum and urine) after bisphosphonate initiation is considered a reasonably reliable surrogate of bisphosphonate efficacy and potency. Maximum suppression of bone resorption occurs within approximately 3 months of initiation of oral bisphosphonate therapy given daily, weekly, or monthly and remains roughly constant with continuation of treatment.10–12 Resorption is suppressed more rapidly after intravenous (IV) bisphosphonate administration than after oral bisphosphonate therapy.
As might be anticipated, length of suppression is largely a function of bisphosphonate potency for mineral matrix binding, such that the most potent bisphosphonate, zoledronic acid, at a dose of either 4 mg13 or 5 mg (the dose approved by the Food and Drug Administration [FDA] for osteoporosis),14 effectively suppresses biochemical markers of bone resorption for up to 1 year in women with postmenopausal osteoporosis. Although the precise biologic half-lives of the currently used nitrogen-containing bisphosphonates remain the subject of debate largely because of technical challenges required to determine bisphosphonate levels in urine and serum, estimates for the potent bisphosphonate alendronate suggest a biologic half-life of more than 10 years after single-dose IV administration.15
A critical feature governing the clinical pharmacology of bisphosphonates is their bioavailability. As a class, bisphosphonates are very hydrophilic. Accordingly, they are poorly absorbed from the gastrointestinal tract after oral administration (generally with absorption of <1% for an oral dose), instead undergoing paracellular transport because they are not lipophilic.16 Further, only about 50% of the absorbed drug is selectively retained in the skeleton, whereas the remainder is eliminated in the urine without being metabolized. Skeletal uptake and retention are primarily dependent on host factors (renal function, prevalent rate of bone turnover, and binding site availability) and bisphosphonate potency for bone matrix.12 The amount of bisphosphonate retained after either oral or IV administration varies widely both between patients and across clinical conditions and is primarily believed to reflect variations in bone turnover.12
A previous impediment for many patients prescribed oral bisphosphonate therapy was the inconvenience associated with daily oral administration (requiring patients to remain upright for 30 minutes and refrain from eating any food both 2 hours before and at least 30 minutes after pill ingestion) and the relatively common association with gastrointestinal symptoms. The more recent development of pharmacologically equivalent preparations allowing for once-weekly (alendronate or risedronate) or even monthly (ibandronate or risedronate) oral administration has profoundly affected bisphosphonate delivery for most patients for whom convenience (and thus adherence to therapy) was an issue and has correspondingly lead to higher rates of adherence.17,18 Further, the availability of IV preparations (pamidronate, ibandronate, and zoledronic acid), which for most clinical conditions require even less frequent dosing, has eliminated the gastrointestinal adverse effects incurred by some patients managed with oral bisphosphonates, although the rate of acute phase reactions characterized by flulike symptoms (low-grade fever, myalgias and arthralgias, or headache) is increased in patients receiving IV rather than oral bisphosphonate treatment.14
Role in Clinical Practice
As aforementioned, bisphosphonates promote the apoptosis of osteoclasts actively engaged in the degradation of mineral on the bone surface. Accordingly, bisphosphonates have become the primary therapy for managing skeletal conditions characterized by increased osteoclast-mediated bone resorption. Such excessive resorption underlies several pathologic conditions for which bisphosphonates are now commonly used, including multiple forms of osteoporosis (juvenile, postmenopausal or involutional [senile], glucocorticoid-induced, transplant-induced, immobility-induced, and androgen-deprivation–related), Paget disease of bone, osteogenesis imperfecta (OI), hypercalcemia, and malignancy metastatic to bone.
Although each of the nitrogen-containing bisphosphonates is more potent than the non–nitrogen-containing bisphosphonates, their ability to suppress osteoclast activity (as measured by biochemical markers of bone turnover) varies. However, whether superior suppression of bone turnover is relevant for fracture prevention remains to be determined. Indeed, data suggest that adherence to long-term bisphosphonate therapy, rather than the specific bisphosphonate used, is the most important factor in determining the effectiveness of treatment for limiting fracture risk.19,20 Accordingly, studies examining bisphosphonate therapy adherence suggest that, by addressing patient concerns of medication safety and timing, clinicians can significantly improve adherence.21 Whether weekly or monthly oral bisphosphonate dosing leads to higher rates of adherence to therapy is currently unknown.
Osteoporosis
The most common clinical condition for which bisphosphonate therapy is used is osteoporosis, a skeletal condition characterized by compromised bone strength resulting in an increased risk of fracture. As previously noted, osteoporosis is a clinically heterogeneous disease with a range of origins, including hormone loss (postmenopausal and androgen-deprivation), iatrogenic (glucocorticoid-induced and transplant-related), physical (immobility), and genetic (eg, juvenile and OI-associated). Often these conditions overlap within individual patients.
Postmenopausal osteoporosis is characterized by an imbalance between osteoclast-mediated bone resorption and osteoblast-mediated bone formation such that bone resorption is increased. This relative imbalance leads to diminution of skeletal mass, deterioration of bone microarchitecture, and increased fracture risk. During the past 2 decades, bisphosphonate therapy has become the leading clinical intervention for postmenopausal osteoporosis because of the ability of bisphosphonates to selectively suppress osteoclast activity and thereby retard bone resorption. The fracture reduction and concomitant increases in bone density generally seen with bisphosphonate use are believed to result from a decline in the activation frequency of new remodeling units formed by osteoclasts, with relative preservation (at least initially) of osteoblast activity. As such, the initial stabilization and retention of trabecular connectivity allow the duration of secondary mineral deposition on the structural scaffold to be prolonged, thereby increasing the percentage of bone structural units that reach a maximum degree of mineralization.22 This increase in the mean degree of skeletal mineralization underlies both improvements in bone density and reductions in fracture risk after bisphosphonate therapy.
Importantly, this role for bisphosphonates was indirectly buttressed by the early termination of the estrogen and progesterone arm of the Women’s Health Initiative (WHI), because of concern about increased rates of coronary artery disease and breast cancer among women receiving hormonal therapy. For most practitioners and patients, the WHI results effectively limited the practice of treating postmenopausal osteoporosis with hormone replacement therapy, despite the strong evidence provided in the WHI and previous studies that estrogen is highly effective in preventing fractures.23
Among the oral bisphosphonates, both alendronate and risedronate have been conclusively demonstrated to reduce the number of vertebral24–26 and hip fractures,24,27 progression of vertebral deformities, and height loss in postmenopausal women with osteoporosis.28 Ibandronate, developed more recently and available in both oral and IV preparations, has been demonstrated to reduce only the risk of vertebral fracture,29,30 although the sample size estimates used did not allow sufficient power to detect an effect on nonvertebral or hip fractures. The relative fracture risk reduction in vertebral, hip, and nonvertebral sites in post-menopausal women with known osteoporosis after 3 years of bisphosphonate treatment is compared in the Table.

Reductions in fracture incidence occur before demonstrable changes (measured by dual-energy x-ray absorptiometry [DXA]) in bone mineral density (BMD), suggesting that stabilization of existing skeletal microarchitecture or decreased bone turnover is sufficient for fracture risk reduction.31 Daily alendronate use at doses of 10 mg for up to 10 years was well tolerated and was not associated with adverse skeletal outcomes.32 Whereas nearly all osteoporosis trials in which bisphosphonate therapy has been used involved postmenopausal women, general trials that have examined men with a diagnosis of either low bone mass or osteoporosis have demonstrated similar responses to bisphosphonate therapy.33–35
In the Fracture Intervention Trial Long-term Extension, postmenopausal women with low femoral neck BMD (but not necessarily with DXA-defined osteoporosis) were treated with daily alendronate for 5 years and then randomized to receive either alendronate or placebo for an additional 5 years. Women who discontinued alendronate therapy had statistically significant, although clinically relatively small, declines in BMD and associated increases in biochemical markers of bone turnover compared with women who continued therapy.36 Importantly, no significant differences were found for either nonvertebral fractures or all clinical fractures; however, there was a slightly higher (and statistically significant) risk of clinical vertebral fractures in the placebo group (absolute risk, 2.9%), but this was not a primary or secondary study end point. Formal studies of alendronate cessation with more statistical power for fracture assessment after discontinuation as a primary end point or of other bisphosphonates have not yet established that, for at least some patients with postmenopausal osteoporosis, a drug holiday could be reasonable after a period of bisphosphonate therapy.
Initial studies used daily bisphosphonate dosing; more recent studies have focused on weekly (alendronate and risedronate) or monthly (ibandronate, and more recently risedronate37) dosing, regimens believed to have pharmacodynamic equivalence to daily dosing of each drug. However, all studies to date using intermittent weekly or monthly oral bisphosphonate therapy have relied on surrogate markers, such as biochemical markers of bone resorption or changes in BMD measured by DXA, rather than primary fracture outcomes, for determination of efficacy. In contrast, the BONE trial, in which oral ibandronate was administered every other day for 12 doses every 3 months, did reduce vertebral fractures with intermittent dosing,30 although this dosing regimen is not approved by the FDA for treatment of postmenopausal osteoporosis. Nonetheless, intermittent weekly or monthly therapy is believed to be biologically equivalent for fracture prevention and has become the standard of care.
More recently, both ibandronate and zoledronic acid have been approved for IV administration to treat postmenopausal osteoporosis. Whereas ibandronate is approved for quarterly administration, zoledronic acid is approved for once-yearly administration. During the 3-year Health Outcomes and Reduced Incidence with Zoledronic Acid Once Yearly (HORIZON) study period, annual IV administration of zoledronic acid led to significant decreases in vertebral (70% reduction), hip (41% reduction), and nonvertebral (25% reduction) fractures, with significant increases in BMD at the lumbar spine, hip, and femoral neck.14 In addition, administration of IV zoledronic acid within 90 days of surgical hip fracture repair and yearly thereafter was recently shown to reduce the incidence of any new clinical fracture by 35% and was associated with a 28% reduction in mortality.38 Further, in patients who have been treated with weekly alendronate for at least 1 year, switching to yearly zoledronic acid was not inferior to alendronate continuation, but yearly administration was preferred by patients.39 Whether IV preparations will become preferred bisphosphonate formulations for management of postmenopausal osteoporosis or after hip fracture is unknown. Nonetheless, it is clear that IV bisphosphonate delivery is particularly useful if adherence or gastrointestinal tolerance is a barrier to oral therapy or if patients prefer the relative convenience of IV bisphosphonate therapy.
Finally, several studies have focused on optimal timing of bisphosphonate therapy for management of osteoporosis in conjunction with other pharmacological agents with skeletal activity. Although combining a bisphosphonate with either estrogen or the selective estrogen-receptor modulator raloxifene leads to a slightly greater increase in BMD than treatment with a bisphosphonate alone, no good clinical trial data on fracture rates support routine use of these combinations.40,41 Other studies have evaluated patients receiving either recombinant full-length 1–84 human parathyroid hormone (PTH) or the PTH fragment 1–34 (teriparatide).42–44 In general, prior bisphosphonate treatment appears to blunt the PTH-induced anabolic skeletal response, as does concomitant treatment using bisphosphonate and either PTH or teriparatide.45,46 The most robust skeletal anabolic effects are seen in patients who receive initial PTH treatment and are subsequently maintained with bisphosphonate therapy.35,47,48
Glucocorticoid-Induced and Transplant-Associated Osteoporosis
Whereas bisphosphonates have become the primary therapeutic choice for treatment of postmenopausal osteoporosis, few recognize that glucocorticoid therapy leads to bone loss. A recent study found that most patients receiving long-term glucocorticoid therapy received neither regular BMD assessment nor a prescription for any medication for osteoporosis management.49 Numerous clinical trials have now determined that bisphosphonates are highly effective at limiting bone losses in patients receiving glucocorticoids or transplants. Recent work has shown that, in patients receiving a daily dose of at least 7.5 mg of prednisone, alendronate prevented bone loss more effectively than did the vitamin D3 analogue alfacalcidol.50 Further, in glucocorticoid-treated patients at high risk of fracture, including those with a history of fractures, those with rheumatoid arthritis, or those receiving high doses of glucocorticoid, bisphosphonate therapy is cost-effective.51
Accordingly, risedronate has been approved in the United States for both prevention and treatment of GIO and alendronate for the treatment of GIO. Both are more effective when calcium intake and vitamin D intake are adequate. As well, IV treatment with either pamidronate or ibandronate has been shown to limit skeletal loss from glucocorticoid therapy,52,53 although neither is yet approved for this indication. Notably, multiple studies have documented that both oral and IV bisphosphonate therapy are capable of limiting the bone loss that frequently occurs with either solid organ54–58 or bone marrow transplant.59–62
Finally, a recent study showed that patients with GIO treated with teriparatide had a greater increase in lumbar spine BMD and fewer new vertebral fractures than did patients who received daily alendronate during the course of 18 months.63 Whether teriparatide should supplant bisphosphonate therapy as the treatment of choice for patients with established osteoporosis who are receiving long-term glucocorticoid therapy remains unknown.
Immobility-Induced Osteoporosis and Other Causes of Acute Bone Loss
Immobilized patients, such as those with a recent spinal cord injury or cerebrovascular event, undergo rapid loss of bone, leading to a substantially increased risk of fracture, hypercalcemia, and frequently nephrolithiasis. Both oral (alendronate)64 and IV (pamidronate)65 bisphosphonate therapy have been shown to attenuate this bone loss and reduce biochemical markers of bone resorption. However, the number of clinical trials conducted using both these drugs remains small. Thus, fracture incidence, rates of nephrolithiasis, and long-term safety remain to be determined.
Unlike the generalized bone loss that occurs after immobilization, acute localized periprosthetic bone loss with associated implant loosening is a frequent complication in patients who undergo cementless total hip arthroplasty. Both alendronate66 and risedronate67 attenuate this acute periprosthetic bone loss of the proximal femur, although the long-term effect of bisphosphonate treatment on maintenance of implant integrity has not yet been reported.
Paget Disease of Bone
Whereas postmenopausal osteoporosis is characterized by generalized bone loss from increased osteoclast activity, Paget disease of bone involves 1 or more areas of disordered bone remodeling, in which accelerated osteoclast-mediated bone resorption is followed by imperfect osteoblast-mediated bone deposition.68 The resulting mix of poorly formed woven and lamellar bone frequently results in pain, fractures, and serious deformity, including bowing of weight-bearing long bones, skull enlargement, or numerous other skeletal deformities. As the cornerstone of therapy for Paget disease of bone, bisphosphonates profoundly suppress the increased bone resorption underlying the disease, generally leading to normalization of serum alkaline phosphatase levels used to monitor disease activity. Oral (alendronate69 and risedronate70) and IV (pamidronate71 and the recently approved zoledronic acid72) bisphosphonates are all FDA-approved for the treatment of Paget disease of bone and have largely replaced earlier FDA-approved therapies (
Bisphosphonates in Malignancy
Many cancers are osteotropic and either metastasize to the skeleton (including but not limited to primary malignancies of the breast, prostate, lung, or kidney) or grow primarily within the bone marrow (multiple myeloma), where this growth frequently leads to hypercalcemia, severe bone pain, skeletal destruction, and pathologic fractures. Indeed, the skeleton is the most common site of metastatic disease, and 90% or more of patients with advanced cancer develop skeletal lesions.73
Breast Cancer
For patients with breast cancer metastatic to bone, treatment with IV preparations of pamidronate,74–76 zoledronic
Whether bisphosphonate use has an adjunct role in the treatment of women with breast cancer but no evidence of skeletal metastases is currently unknown but is suggested by the provocative finding that women with clinically limited operable breast cancer who received clodronate for 2 years had statistically significant reductions in development of bone metastases while receiving bisphosphonate therapy, as well as reductions in overall mortality when they were followed up for 6 years.82 Although bisphosphonate therapy for women receiving hormonal treatment of breast cancer has received less attention, the important role of limiting bone turnover to maintain skeletal integrity (particularly among premenopausal women in whom pharmacological estrogen deficiency has been introduced) has been more recently appreciated.83 Optimal bisphosphonate management strategies corresponding to numerous available pharmacological ovarian ablation regimens remain to be determined, although zoledronic acid (4 mg IV given every 6 months)84 has recently been demonstrated to prevent bone loss in premenopausal women receiving endocrine-based therapy for hormone-sensitive breast cancer. Likewise, in postmenopausal women with early hormone-dependent breast cancer, weekly oral risedronate was recently shown to prevent bone loss in those receiving aromatase inhibitor therapy.85
Prostate Cancer
Breast cancer is characterized by osteolytic lesions, but skeletal metastases from prostate cancer have been described as osteoblastic. The role of increased bone resorption in metastatic prostate cancer has recently been recognized.86 Among the bisphosphonates, only zoledronic acid has been demonstrated to reduce skeletal
As with women who undergo chemical hormonal ablation, men with hormone-responsive prostate cancer who receive androgen-deprivation therapy can benefit from judicious bisphosphonate use. Whereas IV pamidronate therapy prevented bone loss at both the hip and the spine in men with nonmetastatic prostate cancer who received gonadotropin-releasing hormone agonist therapy,89 a single annual dose of IV zoledronic acid was recently demonstrated to lead to increases in both spine and hip BMD (rather than the declines seen in patients who received placebo). These results demonstrate that annual IV bisphosphonate treatment can be a useful adjunct to maintain skeletal integrity in androgen-deprived men90 and are similar to results obtained with a more frequent dosing schedule.91 Oral risedronate at a daily dosage of 2.5 mg has also recently been shown to prevent BMD loss at the hip and been associated with a 4.9% increase at the lumbar spine.92
Multiple Myeloma
In multiple myeloma, clonal proliferation of malignant plasma cells within the bone marrow cavity results in osteolysis and skeletal destruction, accounting for much of the morbidity associated with the disease. Multiple studies have shown that both pamidronate and zoledronic acid have an important palliative role in reducing the incidence of hypercalcemia and skeletal
Given that patients with multiple myeloma have the highest incidence of ONJ among all oncology patients receiving bisphosphonate therapy, the choice of bisphosphonate, dosage, and duration of therapy have been the focus of considerable debate, cumulating in clinical practice guidelines from the American Society of Clinical Oncology96 and, more recently, a consensus statement from the Mayo Clinic Myeloma Group97 on the basis of a comprehensive review of the evolving literature. In the Mayo consensus statement, monthly infusion of pamidronate (because of a perceived higher risk of ONJ in patients receiving zoledronic acid) was favored, with discontinuation after 2 years if patients achieve remission and require no further myeloma treatment. If active treatment is still required, pamidronate can be continued at a reduced schedule of every 3 months. Although the International Myeloma Working Group generally agreed with the Mayo consensus statement, the group suggested that pamidronate therapy could be discontinued after a patient is in 1 year of clinical remission and that a reduced dosing schedule was not indicated.98 Thus, although bisphosphonates remain an important aspect of the pharmacological approach to myeloma bone disease, questions regarding their optimal use remain.
Other Malignancies
Use of bisphosphonates in other malignancies less frequently metastatic to bone, such as renal cell carcinoma, has been demonstrated to delay the onset and progression of
Bisphosphonate Therapy for Children
Although bisphosphonates have been used most extensively in adults, during the past decade they have become the mainstay of therapy for OI, a heritable skeletal disorder characterized by substantially diminished bone mass and severe fragility, usually resulting from mutations in the genes for type I collagen. A regimen developed by Glorieux100 of cyclic IV pamidronate (given in 3-day cycles every 2 to 4 months at an annual dose of 9 mg/kg) has been used most successfully, leading to an 88% increase in cortical thickness, a 46% increase in trabecular bone volume,101 and substantial improvement in functional status. More recently, several studies have demonstrated that oral alendronate can also lead to substantial increases in BMD and can limit fractures in OI affecting children.102–104 Although the precise mechanism by which bisphosphonates limit fractures in OI is unknown, histomorphometric analyses of bone biopsy specimens from patients with OI demonstrate increased rates of bone turnover resulting from increased osteoclast relative to osteoblast activity, leading to an overall loss of bone with each remodeling cycle.105 By specifically inhibiting osteoclast-mediated bone resorption, bisphosphonates presumptively allow bone-forming osteoblasts more time to promote bone formation, albeit in the setting of abnormal collagen matrix. Indeed, histomorphometric analyses of iliac crest biopsy specimens from patients with OI who had received pamidronate therapy demonstrated increased cortical thickness and number of trabeculae but no increase in trabecular thickness.101,106
Although bisphosphonate treatment is well established for OI in children, data are limited on efficacy and on risk of harm when bisphosphonates are used in children with osteoporosis secondary to chronic illness (such as cystic fibrosis, juvenile rheumatoid arthritis, or anorexia nervosa) or in those who have had serious burns. A recent systematic review of bisphosphonate therapy for children and adolescents with secondary osteoporosis concluded that too little evidence is available to support bisphosphonates as standard therapy, although treatment for periods of 3 years or less appears to be well tolerated.107 Well-constructed studies are required to develop clear guidelines to diagnose and treat all forms of osteoporosis in children.108
Finally, given the long skeletal half-life of bisphosphonates and evidence that pamidronate can be found in urine specimens up to 8 years after administration,109 care is warranted when considering bisphosphonate treatment for either adolescent or young girls who will reach reproductive maturity within a decade of treatment. At present, only limited, anecdotal data have assessed the safety of long-term pamidronate110 or other bisphosphonate treatment during fetal development.

Bisphosphonates in clinical practice are utilized to treat osteoporosis, Paget’s disease of the bone, bone metastasis, multiple myeloma, and other health issues with fragile bones. Although bisphosphonates are recommended as one of the first-line treatments for post-menopausal osteoporosis, research studies have previously discussed the adverse effects of this class of drug/medication. It’s essential for patients to talk to their healthcare professional regarding the treatment options for their injuries and/or conditions.
Dr. Alex Jimenez D.C., C.C.S.T. Insight
Clinical Concerns Associated with Bisphosphonate Therapy
Osteonecrosis of the Jaw
Among potential adverse clinical events associated with the use of bisphosphonates, none has received greater attention than ONJ. As reviewed by Woo et al,111 nearly all ONJ cases (94%) have been described in patients receiving high doses of IV bisphosphonates (primarily zoledronic acid and pamidronate) for oncologic conditions. Prevalence
Whereas the incidence of ONJ is estimated to be 1 to 10 per 100 oncology patients, the risk of ONJ appears to be substantially lower among patients receiving oral bisphosphonate therapy for osteoporosis, with an estimated incidence of approximately 1 in 10,000 to 1 in 100,000 patient treatment years, although this estimate is based on incomplete data.114 Associated risk factors appear to be poor oral hygiene, a history of dental procedures or denture use, and prolonged exposure to high IV bisphosphonate doses.115,116 Whether concomitant chemotherapy or glucocorticoid use leads to an increased risk of ONJ is unknown.117 Once established, care for ONJ is largely supportive, with antiseptic oral rinses, antibiotics, and limited surgical debridement as necessary leading to healing in most cases.118 Although evidence-based guidelines at this time have not been established for any single malignancy or bisphosphonate, careful attention to dental hygiene including an oral cavity examination for active or anticipated dental issues, both before bisphosphonate initiation and throughout treatment, is likely to be paramount.
Although use of bisphosphonates and development of ONJ have been temporally associated, a causal relationship has not been identified. Thus, despite the burgeoning scientific literature that has developed since the association between bisphosphonate therapy and ONJ was first reported in 2003,119 many fundamental questions remain unanswered. As a first step in this process, a task force convened by the American Society for Bone and Mineral Research recently provided a standardized definition of ONJ as the presence of exposed bone in the maxillofacial region that does not heal within 8 weeks after identification by a health care professional.114 Given the current paucity of information on the true incidence, risk factors, and clinical approach to both prevention and treatment, preclinical basic and animal studies, as well as well-designed clinical trials, are necessary to both identify patients at increased risk of development of ONJ and more fully understand the association between bisphosphonate therapy and ONJ.
Atrial Fibrillation
In addition to the concern for ONJ, another concern with bisphosphonate therapy, which has recently come to light, is atrial fibrillation. In the HORIZON Pivotal Fracture Trial, in which patients were treated annually with IV zoledronic acid, a statistically significant increase in the incidence of serious atrial fibrillation (defined as events resulting in hospitalization or disability or judged to be life-threatening) was noted.14 The etiology of this electrophysiologic abnormality is unknown. Whether other bisphosphonate preparations are associated with increased rates of atrial fibrillation is currently unknown, but recent post hoc analysis of data from the pivotal Fracture Intervention Trials120 and from a large population-based case-control study121 suggest a correlation between alendronate administration and a slightly increased incidence of atrial fibrillation, although a larger population-based case-control study showed no evidence of an increased risk of atrial fibrillation or flutter with alendronate use.122 To date, concerns for atrial fibrillation do not appear to extend to patients receiving risedronate,123 nor was an increased rate of atrial fibrillation seen in the HORIZON Recurrent Fracture Trial, in which patients received IV zoledronic acid after a hip fracture.38 Clearly, more studies examining the potential relationship between bisphosphonate use and atrial fibrillation are warranted, as are focused discussions between clinicians and patients either currently managed with or considering initiation of bisphosphonate treatment.
Oversuppression of Bone Turnover
Because bisphosphonates inhibit osteoclast activity, there has been some concern that prolonged bisphosphonate treatment leads to “frozen bone,” characterized by over-suppression of bone remodeling, an impaired ability to repair skeletal microfractures, and increased skeletal fragility. Although increased rates of microfractures have been found in dogs treated with high doses of bisphosphonates,124 this finding does not appear to be common among postmenopausal women with osteoporosis treated with either oral or IV bisphosphonate therapy,22,125 although isolated cases of severely suppressed bone turnover and associated fractures have been reported.126,127 Nonetheless, the optimal duration of bisphosphonate therapy for postmenopausal osteoporosis, and nearly all other conditions for which bisphosphonates are used, remains unclear.
Hypocalcemia
Hypocalcemia after bisphosphonate administration most frequently follows IV infusion and can occur in patients with high rates of osteoclast-mediated bone resorption (such as in patients with either Paget disease of bone128 or a substantial skeletal tumor burden129), previously unrecognized hypoparathyroidism,130 impaired renal function, or hypovitaminosis D before treatment.131 Treatment is largely supportive, with calcium and vitamin D supplements as appropriate.
Acute Inflammatory Response
Approximately 10% to 30% of patients receiving their first nitrogen-containing bisphosphonate infusion will experience an acute phase reaction, most commonly characterized by transient pyrexia with associated myalgias, arthralgias, headaches, and influenza-like symptoms. This rate declines by more than half with each subsequent infusion, such that a rate of 2.8% was found after the third infusion in the HORIZON trial.14 The acute phase response is believed to be the result of proinflammatory cytokine production by peripheral blood ?? T cells.132 Pretreatment with histamine receptor antagonists or antipyretics can reduce the incidence and severity of symptoms among susceptible patients. Occasionally corticosteroids are of benefit.
A relatively rare adverse effect of bisphosphonate therapy of which physicians should be aware is ocular inflammation (conjunctivitis, uveitis, episcleritis, and scleritis). This complication has been found to occur with both oral and IV bisphosphonate therapy. In the largest retrospective study to date, an incidence of approximately 0.1% was found in patients treated with oral risedronate.133 Fortunately, ocular symptoms usually resolve within a few weeks after bisphosphonate discontinuation.
Severe Musculoskeletal Pain
Although all oral and IV bisphosphonate preparations list musculoskeletal pain as a potential adverse effect in their prescribing information, the US FDA recently issued an alert highlighting the possibility of severe, incapacitating musculoskeletal pain that can occur at any point after initiation of bisphosphonate therapy.134 This severe musculoskeletal pain was distinct from the acute phase response described previously. Fewer than 120 cases had been reported by late 2002 for alendronate and mid-2003 for risedronate in total.135 At this time, both risk factors for and incidence of this adverse effect are unknown.
Other Potential Complications of Bisphosphonate Therapy
Other complications associated with the use of oral and IV bisphosphonate therapies are well recognized. Esophageal irritation and erosion can occur with oral bisphosphonate therapy, particularly in patients with known gastroesophageal reflux disease or esophageal stricture. Strict maintenance of an upright posture for 30 to 60 minutes after ingestion with a full glass of water, depending on the oral bisphosphonate, and the use of weekly rather than daily preparations are both likely to limit the risk of adverse effects. For patients unable to tolerate oral bisphosphonates, IV preparations (as noted previously) are now FDA approved and not associated with gastroesophageal irritation.
Bisphosphonate doses and infusion rates should be adjusted for patients with moderate to severe renal insufficiency. If used in patients with creatine clearance values lower than 30 mL/min, bisphosphonates must be used cautiously. Particularly in patients who receive IV preparations, bisphosphonates can lead to rapid deterioration of renal function,136,137 likely because of their local accumulation in the kidney. For patients with renal insufficiency who receive IV bisphosphonate therapy, renal function both before and after drug administration should be determined. In patients with mild to moderate renal impairment, oral bisphosphonates rarely lead to further deterioration in renal function, likely because of their poor absorption across the gastrointestinal tract and thus limited short-term bioavailability.
Unresolved Questions
Bisphosphonates have been and continue to be used for other conditions without an FDA-approved indication for therapy. As noted, these include various pediatric populations with low bone mass, incident fractures, and prolonged immobility. Many healthy premenopausal women with either radiographic osteopenia or osteoporosis without fractures and postmenopausal women with osteopenia but without fractures now receive bisphosphonate therapy. Until further studies address these important clinical questions, it is important to tell such patients that we currently lack sufficient data from well-controlled clinical trials to determine either benefits or risks assumed with these pharmacological interventions.
Role of Calcium and Vitamin D
Despite the good intentions of many practitioners to limit fractures in their patients by instituting bisphosphonate therapy, the importance of assuring adequate vitamin D and calcium intake both before and after starting bisphosphate therapy is frequently overlooked. Hypovitaminosis D is common among many patient populations that are also prescribed bisphosphonate therapy and is particularly common among elderly patients who frequently have limited sun exposure, reduced dietary intake, or some renal impairment. This vitamin D insufficiency or deficiency limits dietary absorption of calcium, leading to secondary hyperparathyroidism and loss of skeletal calcium to maintain normocalcemia. Accordingly, among elderly women with osteoporosis, the persistence of secondary hyperparathyroidism blunted the increase in BMD in the lumbar spine in response to weekly alendronate.138 Although currently available data offer no consensus on optimal serum levels of 25-hydroxyvitamin D, a level of 30 ng/mL (75 nmol/L) or more is generally considered to be adequate; vitamin D intoxication occurs only when levels are higher than 150 ng/mL (374 nmol/L).139 For a more complete review of the role of vitamin D in maintenance of skeletal health and for recommendations for vitamin D replacement, please refer to the excellent recent review by Holick.139
Although guidelines for the maintenance of optimal vitamin D levels have changed substantially as we appreciate that vitamin D insufficiency and deficiency affect a far greater proportion of the population than previously recognized, recommendations for optimal calcium intake have been modified only slightly since being addressed by an expert panel convened by the National Institutes of Health in 1994.140 The panel concluded that optimal calcium intake is estimated to be 1000 mg/d for both premenopausal and postmenopausal women receiving estrogen replacement therapy and 1500 mg/d for postmenopausal women not receiving estrogen. Men younger than 65 years were estimated to require 1000 mg/d of calcium and men older than 65 years to require 1500 mg/d.140 More recent recommendations from the National Osteoporosis Foundation have suggested a calcium intake of 1000 mg/d for both men and women younger than 50 years, with an increase to 1200 mg/d from age 50 years onward.141 These recommendations are consistent with those of the Food and Nutrition Board of the Institute of Medicine.142 Further recommendations for calcium intake in children are detailed in both the National Institutes of Health’s and Institute of Medicine’s guidelines.140,142
Conclusion
Since their introduction to clinical practice, bisphosphonates have transformed the clinical care of an array of skeletal disorders characterized by excessive osteoclast-mediated bone resorption. Accordingly, the informed and judicious use of bisphosphonates confers a clear clinical benefit for carefully selected patients that
Acknowledgments
We thank James M. Peterson for assistance with the figures.
Preparation of this article was supported by a Mayo Career Development Award to Dr Drake.
Dr Khosla has received research support from Procter & Gamble and has served on the advisory board for Novartis.
Glossary
- ATP – adenosine triphosphate
- BMD – bone mineral density
- DXA – dual-energy x-ray absorptiometry
- FDA – Food and Drug Administration
- GIO – glucocorticoid-induced osteoporosis
- HORIZON – Health Outcomes and Reduced Incidence with Zoledronic Acid Once Yearly
- IV – intravenous
- OI – osteogenesis imperfecta
- ONJ – osteonecrosis of the jaw
- PPi – inorganic pyrophosphate
- PTH – parathyroid hormone
- WHI – Women’s Health Initiative
Footnotes
Individual reprints of this article are not available.
According to the article above, although the utilization of bisphosphonates in clinical practice provides healthcare professionals with new treatment options for skeletal disorders, further research studies are still required. Information referenced from the National Center for Biotechnology Information (NCBI). The scope of our information is limited to chiropractic as well as to spinal injuries and conditions. To discuss the subject matter, please feel free to ask Dr. Jimenez or contact us at
Curated by Dr. Alex Jimenez
1. Russell RG. Bisphosphonates: from bench to bedside. Ann N Y Acad Sci. 2006;1068:367–401. [PubMed]
2. Fleisch H, Russell RG, Straumann F. Effect of pyrophosphate on hydroxyapatite and its implications in calcium homeostasis. Nature. 1966;212(5065):901–903. [PubMed]
3. Russell RG, Muhlbauer RC, Bisaz S, Williams DA, Fleisch H. The influence of pyrophosphate, condensed phosphates, phosphonates and other phosphate compounds on the dissolution of hydroxyapatite in vitro and on bone resorption induced by parathyroid hormone in tissue culture and in thyroparathyroidectomised rats. Calcif Tissue Res. 1970;6(3):183–196. [PubMed]
4. Plotkin LI, Weinstein RS, Parfitt AM, Roberson PK, Manolagas SC, Bellido T. Prevention of osteocyte and osteoblast apoptosis by bisphosphonates and calcitonin. J Clin Invest. 1999;104(10):1363–1374. [PMC free article] [PubMed]
5. Plotkin LI, Aguirre JI, Kousteni S, Manolagas SC, Bellido T. Bisphosphonates and estrogens inhibit osteocyte apoptosis via distinct molecular mechanisms downstream of extracellular signal-regulated kinase activation. J Biol Chem. 2005 Feb 25;280:7317–7325. Epub 2004 Dec 6. [PubMed]
6. Dunford JE, Thompson K, Coxon FP, et al. Structure-activity relationships for inhibition of farnesyl diphosphate synthase in vitro and inhibition of bone resorption in vivo by nitrogen-containing bisphosphonates. J Pharmacol Exp Ther. 2001;296(2):235–242. [PubMed]
7. Kavanagh KL, Guo K, Dunford JE, et al. The molecular mechanism of nitrogen-containing bisphosphonates as antiosteoporosis drugs. Proc Natl Acad Sci U S A. 2006 May 16;103(20):7829–7834. Epub 2006 May 9. [PMC free article] [PubMed]
8. Hall A. Rho GTPases and the actin cytoskeleton. Science. 1998;279(5350):509–514. [PubMed]
9. Luckman SP, Hughes DE, Coxon FP, Graham R, Russell G, Rogers MJ. Nitrogen-containing bisphosphonates inhibit the mevalonate pathway and prevent post-translational prenylation of GTP-binding proteins, including Ras. J Bone Miner Res. 1998;13(4):581–589. [PubMed]
10. Rizzoli R, Greenspan SL, Bone GIII, et al. Alendronate Once-Weekly Study Group. Two-year results of once-weekly administration of alendronate 70 mg for the treatment of postmenopausal osteoporosis. J Bone Miner Res. 2002;17(11):1988–1996. [PubMed]
11. Brown JP, Kendler DL, McClung MR, et al. The efficacy and tolerability of risedronate once a week for the treatment of postmenopausal osteoporosis. Calcif Tissue Int. 2002 Aug;71(2):103–111. Epub 2002 Jun 27. [PubMed]
12. Cremers SC, Pillai G, Papapoulos SE. Pharmacokinetics/pharmacodynamics of bisphosphonates: use for optimisation of intermittent therapy for osteoporosis. Clin Pharmacokinet. 2005;44(6):551–570. [PubMed]
13. Reid IR, Brown JP, Burckhardt P, et al. Intravenous zoledronic acid in postmenopausal women with low bone mineral density. N Engl J Med. 2002;346(9):653–661. [PubMed]
14. Black DM, Delmas PD, Eastell R, et al. HORIZON Pivotal Fracture Trial. Once-yearly zoledronic acid for treatment of postmenopausal osteoporosis. N Engl J Med. 2007;356(18):1809–1822. [PubMed]
15. Khan SA, Kanis JA, Vasikaran S, et al. Elimination and biochemical responses to intravenous alendronate in postmenopausal osteoporosis. J Bone Miner Res. 1997;12(10):1700–1707. [PubMed]
16. Lin JH. Bisphosphonates: a review of their pharmacokinetic properties. Bone. 1996;18(2):75–85. [PubMed]
17. Penningvan Beest FJ, Goettsch WG, Erkens JA, Herings RM. Determinants of persistence with bisphosphonates: a study in women with postmenopausal osteoporosis. Clin Ther. 2006;28(2):236–242. [PubMed]
18. Rossini M, Bianchi G, Di Munno O, et al. Osteoporosis in Clinical Practice (TOP) Study Group. Determinants of adherence to osteoporosis treatment in clinical practice. Osteoporos Int. 2006;17(6):914–921. Epub 2006 Mar 15. [PubMed]
19. Yood RA, Emani S, Reed JI, Lewis BE, Charpentier M, Lydick E. Compliance with pharmacologic therapy for osteoporosis. Osteoporos Int. 2003 Dec;14(12):965–968. Epub 2003 Sep 19. [PubMed]
20. Siris ES, Harris ST, Rosen CJ, et al. Adherence to bisphosphonate therapy and fracture rates in osteoporotic women: relationship to vertebral and nonvertebral fractures from 2 US claims databases. Mayo Clin Proc. 2006;81(8):1013–1022. [PubMed]
21. Carr AJ, Thompson PW, Cooper C. Factors associated with adherence and persistence to bisphosphonate therapy in osteoporosis: a cross-sectional survey. Osteoporos Int. 2006;17(11):1638–1644. Epub 2006 Aug 1. [PubMed]
22. Boivin GY, Chavassieux PM, Santora AC, Yates J, Meunier PJ. Alendronate increases bone strength by increasing the mean degree of mineralization of bone tissue in osteoporotic women. Bone. 2000;27(5):687–694. [PubMed]
23. Rossouw JE, Anderson GL, Prentice RL, et al. Writing Group for the Women’s Health Initiative Investigators. Risks and benefits of estrogen plus progestin in healthy postmenopausal women: principal results From the Women’s Health Initiative randomized controlled trial. JAMA. 2002;288(3):321–333. [PubMed]
24. Black DM, Cummings SR, Karpf DB, et al. Fracture Intervention Trial Research Group. Randomised trial of effect of alendronate on risk of fracture in women with existing vertebral fractures. Lancet. 1996;348(9041):1535–1541. [PubMed]
25. Cummings SR, Black DM, Thompson DE, et al. Effect of alendronate on risk of fracture in women with low bone density but without vertebral fractures: results from the Fracture Intervention Trial. JAMA. 1998;280(24):2077–2082. [PubMed]
26. Harris ST, Watts NB, Genant HK, et al. Vertebral Efficacy With Risedronate Therapy (VERT) Study Group. Effects of risedronate treatment on vertebral and nonvertebral fractures in women with postmenopausal osteoporosis: a randomized controlled trial. JAMA. 1999;282(14):1344–1352. [PubMed]
27. McClung MR, Geusens P, Miller PD, et al. Hip Intervention Program Study Group. Effect of risedronate on the risk of hip fracture in elderly women. N Engl J Med. 2001;344(5):333–340. [PubMed]
28. Liberman UA, Weiss SR, Broll J, et al. Alendronate Phase III Osteoporosis Treatment Study Group. Effect of oral alendronate on bone mineral density and the incidence of fractures in postmenopausal osteoporosis. N Engl J Med. 1995;333(22):1437–1443. [PubMed]
29. Delmas PD, Recker RR, Chesnut CH, III, et al. Daily and intermittent oral ibandronate normalize bone turnover and provide significant reduction in vertebral fracture risk: results from the BONE study. Osteoporos Int. 2004 Oct;15(10):792–798. Epub 2004 Apr 8. [PubMed]
30. Chesnut IC, III, Skag A, Christiansen C, et al. Oral Ibandronate Osteoporosis Vertebral Fracture Trial in North America and Europe (BONE). Effects of oral ibandronate administered daily or intermittently on fracture risk in postmenopausal osteoporosis. J Bone Miner Res. 2004 Aug;19(8):1241–1249. Epub 2004 Mar 29. [PubMed]
31. Riggs BL, Melton LJ., III Bone turnover matters: the raloxifene treatment paradox of dramatic decreases in vertebral fractures without commensurate increases in bone density [editorial] J Bone Miner Res. 2002;17(1):11–14. [PubMed]
32. Bone HG, Hosking D, Devogelaer JP, et al. Alendronate Phase III Osteoporosis Treatment Study Group. Ten years’ experience with alendronate for osteoporosis in postmenopausal women. N Engl J Med. 2004;350(12):1189–1199. [PubMed]
33. Orwoll E, Ettinger M, Weiss S, et al. Alendronate for the treatment of osteoporosis in men. N Engl J Med. 2000;343(9):604–610. [PubMed]
34. Finkelstein JS, Leder BZ, Burnett SM, et al. Effects of teriparatide, alendronate, or both on bone turnover in osteoporotic men. J Clin Endocrinol Metab. 2006 Aug;91(8):2882–2887. Epub 2006 May 9. [PubMed]
35. Kurland ES, Heller SL, Diamond B, McMahon DJ, Cosman F, Bilezikian JP. The importance of bisphosphonate therapy in maintaining bone mass in men after therapy with teriparatide [human parathyroid hormone(1–34)] Osteoporos Int. 2004 Dec;15(12):992–997. Epub 2004 Jun 3. [PubMed]
36. Black DM, Schwartz AV, Ensrud KE, et al. FLEX Research Group. Effects of continuing or stopping alendronate after 5 years of treatment: the Fracture Intervention Trial Long-term Extension (FLEX): a randomized trial. JAMA. 2006;296(24):2927–2938. [PubMed]
37. Delmas PD, McClung MR, Zanchetta JR, et al. Efficacy and safety of risedronate 150 mg once a month in the treatment of postmenopausal osteoporosis. Bone. 2008 Jan;42(1):36–42. Epub 2004 Jun 3. [PubMed]
38. Lyles KW, Colon-Emeric CS, Magaziner JS, et al. HORIZON Recurrent Fracture Trial. Zoledronic acid and clinical fractures and mortality after hip fracture. N Engl J Med. 2007 Nov 1;357(18):1799–1809. Epub 2007 Sep 17. [PubMed]
39. McClung M, Recker R, Miller P, et al. Intravenous zoledronic acid 5 mg in the treatment of postmenopausal women with low bone density previously treated with alendronate. Bone. 2007 Jul;41(1):122–128. Epub 2007 Mar 24. [PubMed]
40. Greenspan SL, Emkey RD, Bone HG, et al. Significant differential effects of alendronate, estrogen, or combination therapy on the rate of bone loss after discontinuation of treatment of postmenopausal osteoporosis: a randomized, double-blind, placebo-controlled trial. Ann Intern Med. 2002;137(11):875–883. [PubMed]
41. Compston JE, Watts NB. Combination therapy for postmenopausal osteoporosis. Clin Endocrinol (Oxf) 2002;56(5):565–569. [PubMed]
42. Finkelstein JS, Klibanski A, Schaefer EH, Hornstein MD, Schiff I, Neer RM. Parathyroid hormone for the prevention of bone loss induced by estrogen deficiency. N Engl J Med. 1994;331(24):1618–1623. [PubMed]
43. Neer RM, Arnaud CD, Zanchetta JR, et al. Effect of parathyroid hormone (1–34) on fractures and bone mineral density in postmenopausal women with osteoporosis. N Engl J Med. 2001;344(19):1434–1441. [PubMed]
44. Cosman F, Nieves J, Zion M, Woelfert L, Luckey M, Lindsay R. Daily and cyclic parathyroid hormone in women receiving alendronate. N Engl J Med. 2005;353(6):566–575. [PubMed]
45. Black DM, Greenspan SL, Ensrud KE, et al. PaTH Study Investigators. The effects of parathyroid hormone and alendronate alone or in combination in postmenopausal osteoporosis. N Engl J Med. 2003 Sep 25;349(13):1207–1215. Epub 2003 Sep 20. [PubMed]
46. Finkelstein JS, Hayes A, Hunzelman JL, Wyland JJ, Lee H, Neer RM. The effects of parathyroid hormone, alendronate, or both in men with osteoporosis. N Engl J Med. 2003 Sep 25;349(13):1216–1226. Epub 2003 Sep 20. [PubMed]
47. Rittmaster RS, Bolognese M, Ettinger MP, et al. Enhancement of bone mass in osteoporotic women with parathyroid hormone followed by alendronate. J Clin Endocrinol Metab. 2000;85(6):2129–2134. [PubMed]
48. Black DM, Bilezikian JP, Ensrud KE, et al. PaTH Study Investigators. One year of alendronate after one year of parathyroid hormone (1–84) for osteoporosis. N Engl J Med. 2005;353(6):555–565. [PubMed]
49. Curtis JR, Westfall AO, Allison J, et al. Challenges in improving the quality of osteoporosis care for long-term glucocorticoid users: a prospective randomized trial. Arch Intern Med. 2007;167(6):591–596. [PubMed]
50. de Nijs RN, Jacobs JW, Lems WF, et al. STOP Investigators. Alendronate or alfacalcidol in glucocorticoid-induced osteoporosis. N Engl J Med. 2006;355(7):675–684. [PubMed]
51. van Staa TP, Geusens P, Zhang B, Leufkens HG, Boonen A, Cooper C. Individual fracture risk and the cost-effectiveness of bisphosphonates in patients using oral glucocorticoids. Rheumatology (Oxford) 2007 Mar;46(3):460–466. Epub 2006 Aug 9. [PubMed]
52. Boutsen Y, Jamart J, Esselinckx W, Stoffel M, Devogelaer JP. Primary prevention of glucocorticoid-induced osteoporosis with intermittent intravenous pamidronate: a randomized trial. Calcif Tissue Int. 1997;61(4):266–271. [PubMed]
53. Ringe JD, Dorst A, Faber H, Ibach K, Sorenson F. Intermittent intravenous ibandronate injections reduce vertebral fracture risk in corticosteroid-induced osteoporosis: results from a long-term comparative study. Osteoporos Int. 2003 Oct;14(10):801–807. Epub 2003 Aug 28. [PubMed]
54. Krieg MA, Seydoux C, Sandini L, et al. Intravenous pamidronate as treatment for osteoporosis after heart transplantation: a prospective study. Osteoporos Int. 2001;12(2):112–116. [PubMed]
55. Coco M, Glicklich D, Faugere MC, et al. Prevention of bone loss in renal transplant recipients: a prospective, randomized trial of intravenous pamidronate. J Am Soc Nephrol. 2003;14(10):2669–2676. [PubMed]
56. Aris RM, Lester GE, Renner JB, et al. Efficacy of pamidronate for osteoporosis in patients with cystic fibrosis following lung transplantation. Am J Respir Crit Care Med. 2000;162(3 pt 1):941–946. [PubMed]
57. Grotz W, Nagel C, Poeschel D, et al. Effect of ibandronate on bone loss and renal function after kidney transplantation. J Am Soc Nephrol. 2001;12(7):1530–1537. [PubMed]
58. Giannini S, D’Angelo A, Carraro G, et al. Alendronate prevents further bone loss in renal transplant recipients. J Bone Miner Res. 2001;16(11):2111–2117. [PubMed]
59. Tauchmanova L, Ricci P, Serio B, et al. Short-term zoledronic acid treatment increases bone mineral density and marrow clonogenic fibroblast progenitors after allogeneic stem cell transplantation. J Clin Endocrinol Metab. 2005 Feb;90(2):627–634. Epub 2004 Nov 16. [PubMed]
60. Grigg AP, Shuttleworth P, Reynolds J, et al. Pamidronate reduces bone loss after allogeneic stem cell transplantation. J Clin Endocrinol Metab. 2006 Oct;91(10):3835–3843. Epub 2006 Jul 11. [PubMed]
61. D’Souza AB, Grigg AP, Szer J, Ebeling PR. Zoledronic acid prevents bone loss after allogeneic haemopoietic stem cell transplantation. Intern Med J. 2006;36(9):600–603. [PubMed]
62. Yao S, McCarthy PL, Dunford LM, et al. High prevalence of early-onset osteopenia/osteoporosis after allogeneic stem cell transplantation and improvement after bisphosphonate therapy. Bone Marrow Transplant. 2008 Feb;41(4):393–398. Epub 2007 Nov 12. [PubMed]
63. Saag KG, Shane E, Boonen S, et al. Teriparatide or alendronate in glucocorticoid-induced osteoporosis. N Engl J Med. 2007;357(20):2028–2039. [PubMed]
64. Gilchrist NL, Frampton CM, Acland RH, et al. Alendronate prevents bone loss in patients with acute spinal cord injury: a randomized, double-blind, placebo-controlled study. J Clin Endocrinol Metab. 2007 Apr;92(4):1385–1390. Epub 2007 Jan 16. [PubMed]
65. Nance PW, Schryvers O, Leslie W, Ludwig S, Krahn J, Uebelhart D. Intravenous pamidronate attenuates bone density loss after acute spinal cord injury. Arch Phys Med Rehabil. 1999;80(3):243–251. [PubMed]
66. Venesmaa PK, Kroger HP, Miettinen HJ, Jurvelin JS, Suomalainen OT, Alhav EM. Alendronate reduces periprosthetic bone loss after uncemented primary total hip arthroplasty: a prospective randomized study. J Bone Miner Res. 2001;16(11):2126–2131. [PubMed]
67. Yamasaki S, Masuhara K, Yamaguchi K, Nakai T, Fuji T, Seino Y. Risedronate reduces postoperative bone resorption after cementless total hip arthroplasty. Osteoporos Int. 2007 Jul;18(7):1009–1015. Epub 2007 Feb 15. [PubMed]
68. Whyte MP. Paget’s disease of bone. N Engl J Med. 2006;355(6):593–600. [PubMed]
69. Siris E, Weinstein RS, Altman R, et al. Comparative study of alendronate versus etidronate for the treatment of Paget’s disease of bone. J Clin Endocrinol Metab. 1996;81(3):961–967. [PubMed]
70. Siris ES, Chines AA, Altman RD, et al. Risedronate in the treatment of Paget’s disease of bone: an open label, multicenter study. J Bone Miner Res. 1998;13(6):1032–1038. [PubMed]
71. Siris ES. Perspectives: a practical guide to the use of pamidronate in the treatment of Paget’s disease. J Bone Miner Res. 1994;9(3):303–304. [PubMed]
72. Reid IR, Miller P, Lyles K, et al. Comparison of a single infusion of zoledronic acid with risedronate for Paget’s disease. N Engl J Med. 2005;353(9):898–908. [PubMed]
73. Body JJ. Treatment and prevention of bone metastases and myeloma in bone disease. In: Favus MJ, editor. Primer on the Metabolic Diseases and Disorders of Mineral Metabolism. 6. Washington, DC: American Society for Bone and Mineral Research; 2006. pp. 383–390.
74. Hortobagyi GN, Theriault RL, Porter L, et al. Protocol 19 Aredia Breast Cancer Study Group. Efficacy of pamidronate in reducing skeletal complications in patients with breast cancer and lytic bone metastases. N Engl J Med. 1996;335(24):1785–1791. [PubMed]
75. Hortobagyi GN, Theriault RL, Lipton A, et al. Protocol 19 Aredia Breast Cancer Study Group. Long-term prevention of skeletal complications of metastatic breast cancer with pamidronate. J Clin Oncol. 1998;16(6):2038–2044. [PubMed]
76. Lipton A, Theriault RL, Hortobagyi GN, et al. Pamidronate prevents skeletal complications and is effective palliative treatment in women with breast carcinoma and osteolytic bone metastases: long term follow-up of two randomized, placebo-controlled trials. Cancer. 2000;88(5):1082–1090. [PubMed]
77. Rosen LS, Gordon D, Tchekmedyian NS, et al. Long-term efficacy and safety of zoledronic acid in the treatment of skeletal metastases in patients with nonsmall cell lung carcinoma and other solid tumors: a randomized, Phase III, double-blind, placebo-controlled trial. Cancer. 2004;100(12):2613–2621. [PubMed]
78. Clemons MJ, Dranitsaris G, Ooi WS, et al. Phase II trial evaluating the palliative benefit of second-line zoledronic acid in breast cancer patients with either a skeletal-related event or progressive bone metastases despite first-line bisphosphonate therapy. J Clin Oncol. 2006 Oct 20;24(30):4895–4900. Epub 2006 Sep 25. [PubMed]
79. Body JJ, Diel IJ, Lichinitser MR, et al. MF 4265 Study Group. Intravenous ibandronate reduces the incidence of skeletal complications in patients with breast cancer and bone metastases. Ann Oncol. 2003;14(9):1399–1405. [PubMed]
80. Body JJ, Diel IJ, Bell R, et al. Oral ibandronate improves bone pain and preserves quality of life in patients with skeletal metastases due to breast cancer. Pain. 2004;111(3):306–312. [PubMed]
81. Body JJ, Diel IJ, Lichinitzer M, et al. Oral ibandronate reduces the risk of skeletal complications in breast cancer patients with metastatic bone disease: results from two randomised, placebo-controlled phase III studies. Br J Cancer. 2004;90(6):1133–1137. [PMC free article] [PubMed]
82. Powles T, Paterson S, Kanis JA, et al. Randomized, placebo-controlled trial of clodronate in patients with primary operable breast cancer. J Clin Oncol. 2002;20(15):3219–3224. [PubMed]
83. Khan MN, Khan AA. Cancer treatment-related bone loss: a review and synthesis of the literature. Curr Oncol. 2008;15(suppl 1):S30–S40. [PMC free article] [PubMed]
84. Gnant MF, Mlineritsch B, Luschin-Ebengreuth G, et al. Zoledronic acid prevents cancer treatment-induced bone loss in premenopausal women receiving adjuvant endocrine therapy for hormone-responsive breast cancer: a report from the Austrian Breast and Colorectal Cancer Study Group. J Clin Oncol. 2007 Mar 1;25(7):820–828. Epub 2006 Dec 11. [PubMed]
85. Confavreux CB, Fontana A, Guastalla JP, Munoz F, Brun J, Delmas PD. Estrogen-dependent increase in bone turnover and bone loss in postmenopausal women with breast cancer treated with anastrozole: prevention with bisphosphonates. Bone. 2007 Sep;41(3):346–352. Epub 2007 Jun 16. [PubMed]
86. Garnero P, Buchs N, Zekri J, Rizzoli R, Coleman RE, Delmas PD. Markers of bone turnover for the management of patients with bone metastases from prostate cancer. Br J Cancer. 2000;82(4):858–864. [PMC free article] [PubMed]
87. Saad F, Gleason DM, Murray R, et al. Zoledronic Acid Prostate Cancer Study Group. A randomized, placebo-controlled trial of zoledronic acid in patients with hormone-refractory metastatic prostate carcinoma. J Natl Cancer Inst. 2002;94(19):1458–1468. [PubMed]
88. Saad F, Gleason DM, Murray R, et al. Zoledronic Acid Prostate Cancer Study Group. Long-term efficacy of zoledronic acid for the prevention of skeletal complications in patients with metastatic hormone-refractory prostate cancer. J Natl Cancer Inst. 2004;96(11):879–882. [PubMed]
89. Smith MR, McGovern FJ, Zietman AL, et al. Pamidronate to prevent bone loss during androgen-deprivation therapy for prostate cancer. N Engl J Med. 2001;345(13):948–955. [PubMed]
90. Michaelson MD, Kaufman DS, Lee H, et al. Randomized controlled trial of annual zoledronic acid to prevent gonadotropin-releasing hormone agonist-induced bone loss in men with prostate cancer. J Clin Oncol. 2007;25(9):1038–1042. [PMC free article] [PubMed]
91. Smith MR, Eastham J, Gleason DM, Shasha D, Tchekmedyian S, Zinner N. Randomized controlled trial of zoledronic acid to prevent bone loss in men receiving androgen deprivation therapy for nonmetastatic prostate cancer. J Urol. 2003;169(6):2008–2012. [PubMed]
92. Ishizaka K, Machida T, Kobayashi S, Kanbe N, Kitahara S, Yoshida K. Preventive effect of risedronate on bone loss in men receiving androgen-deprivation therapy for prostate cancer. Int J Urol. 2007;14(12):1071–1075. [PubMed]
93. Berenson JR, Lichtenstein A, Porter L, et al. Myeloma Aredia Study Group. Efficacy of pamidronate in reducing skeletal events in patients with advanced multiple myeloma. N Engl J Med. 1996;334:488–493. [PubMed]
94. Berenson JR, Rosen LS, Howell A, et al. Zoledronic acid reduces skeletal-related events in patients with osteolytic metastases [published correction appears in Cancer. 2001;91(10):1956] Cancer. 2001;91(7):1191–1200. [PubMed]
95. Rosen LS, Gordon D, Kaminski M, et al. Zoledronic acid versus pamidronate in the treatment of skeletal metastases in patients with breast cancer or osteolytic lesions of multiple myeloma: a phase III, double-blind, comparative trial. Cancer J. 2001;7(5):377–387. [PubMed]
96. Berenson JR, Hillner BE, Kyle RA, et al. American Society of Clinical Oncology Bisphosphonates Expert Panel. American Society of Clinical Oncology clinical practice guidelines: the role of bisphosphonates in multiple myeloma. J Clin Oncol. 2002;20(17):3719–3736. [PubMed]
97. Lacy MQ, Dispenzieri A, Gertz MA, et al. Mayo Clinic consensus statement for the use of bisphosphonates in multiple myeloma. Mayo Clin Proc. 2006;81(8):1047–1053. [PubMed]
98. Durie BGM. Use of bisphosphonates in multiple myeloma: IMWG response to Mayo Clinic consensus statement [letter] Mayo Clin Proc. 2007;82(4):516–517. [PubMed]
99. Lipton A, Zheng M, Seaman J. Zoledronic acid delays the onset of skeletal-related events and progression of skeletal disease in patients with advanced renal cell carcinoma. Cancer. 2003;98(5):962–969. [PubMed]
100. Glorieux FH. Experience with bisphosphonates in osteogenesis imperfecta. Pediatrics. 2007;119(suppl 2):S163–S165. [PubMed]
101. Rauch F, Travers R, Plotkin H, Glorieux FH. The effects of intravenous pamidronate on the bone tissue of children and adolescents with osteogenesis imperfecta. J Clin Invest. 2002;110(9):1293–1299. [PMC free article] [PubMed]
102. Cho TJ, Choi IH, Chung CY, Yoo WJ, Park MS, Park YK. Efficacy of oral alendronate in children with osteogenesis imperfecta. J Pediatr Orthop. 2005;25(5):607–612. [PubMed]
103. DiMeglio LA, Peacock M. Two-year clinical trial of oral alendronate versus intravenous pamidronate in children with osteogenesis imperfecta. J Bone Miner Res. 2006 Jan;21(1):132–140. Epub 2005 Oct 17. [PubMed]
104. Akcay T, Turan S, Guran T, Bereket A. Alendronate treatment in children with osteogenesis imperfecta. Indian Pediatr. 2008;45(2):105–109. [PubMed]
105. Zeitlin L, Fassier F, Glorieux FH. Modern approach to children with osteogenesis imperfecta. J Pediatr Orthop B. 2003;12(2):77–87. [PubMed]
106. Munns CF, Rauch F, Travers R, Glorieux FH. Effects of intravenous pamidronate treatment in infants with osteogenesis imperfecta: clinical and histomorphometric outcome. J Bone Miner Res. 2005 Jul;20(7):1235–1243. Epub 2005 Feb 21. [PubMed]
107. Ward L, Tricco AC, Phuong P, et al. Bisphosphonate therapy for children and adolescents with secondary osteoporosis. Cochrane Database Syst Rev. 2007;(4):CD005324. [PubMed]
108. Bachrach LK. Consensus and controversy regarding osteoporosis in the pediatric population. Endocr Pract. 2007;13(5):513–520. [PubMed]
109. Papapoulos SE, Cremers SC. Prolonged bisphosphonate release after treatment in children [letter] N Engl J Med. 2007;356(10):1075–1076. [PubMed]
110. Munns CF, Rauch F, Ward L, Glorieux FH. Maternal and fetal outcome after long-term pamidronate treatment before conception: a report of two cases. J Bone Miner Res. 2004 Oct;19(10):1742–1745. Epub 2004 Jul 21. [PubMed]
111. Woo SB, Hellstein JW, Kalmar JR. Narrative [corrected] review: bisphosphonates and osteonecrosis of the jaws [published correction appears in Ann Intern Med. 2006; 145(3):235] Ann Intern Med. 2006;144(10):753–761. [PubMed]
112. Dimopoulos MA, Kastritis E, Anagnostopoulos A, et al. Osteonecrosis of the jaw in patients with multiple myeloma treated with bisphosphonates: evidence of increased risk after treatment with zoledronic acid. Haematologica. 2006 Jul;91(7):968–971. Epub 2006 Jun 1. [PubMed]
113. Corso A, Varettoni M, Zappasodi P, et al. A different schedule of zoledronic acid can reduce the risk of the osteonecrosis of the jaw in patients with multiple myeloma. Leukemia. 2007 Jul;21(7):1545–1548. Epub 2007 Apr 5. [PubMed]
114. Khosla S, Burr D, Cauley J, et al. Bisphosphonate-associated osteonecrosis of the jaw: report of a task force of the American Society for Bone and Mineral Research [editorial] J Bone Miner Res. 2007;22(10):1479–1491. [PubMed]
115. Marx RE, Sawatari Y, Fortin M, Broumand V. Bisphosphonate-induced exposed bone (osteonecrosis/osteopetrosis) of the jaws: risk factors, recognition, prevention, and treatment. J Oral Maxillofac Surg. 2005;63(11):1567–1575. [PubMed]
116. Bamias A, Kastritis E, Bamia C, et al. Osteonecrosis of the jaw in cancer after treatment with bisphosphonates: incidence and risk factors. J Clin Oncol. 2005;23(34):8580–8587. [PubMed]
117. Tosi P, Zamagni E, Cangini D, et al. Osteonecrosis of the jaws in newly diagnosed multiple myeloma patients treated with zoledronic acid and thalidomide-dexamethasone [letter] Blood. 2006;108(12):3951–3952. [PubMed]
118. Bilezikian JP. Osteonecrosis of the jaw—do bisphosphonates pose a risk? N Engl J Med. 2006;355(22):2278–2281. [PubMed]
119. Marx RE. Pamidronate (Aredia) and zoledronate (Zometa) induced avascular necrosis of the jaws: a growing epidemic [letter] J Oral Maxillofac Surg. 2003;61(9):1115–1117. [PubMed]
120. Cummings SR, Schwartz AV, Black DM. Alendronate and atrial fibrillation [letter] N Engl J Med. 2007;356(18):1895–1896. [PubMed]
121. Heckbert SR, Li G, Cummings SR, Smith NL, Psaty BM. Use of alendronate and risk of incident atrial fibrillation in women. Arch Intern Med. 2008;168(8):826–831. [PubMed]
122. Sorensen HT, Christensen S, Mehnert F, et al. Use of bisphosphonates among women and risk of atrial fibrillation and flutter: population based case-control study. BMJ. 2008 Apr 12;7648 12;336:813–816. Epub 2008 Mar 11. [PMC free article] [PubMed]
123. Karam R, Camm J, McClung M. Yearly zoledronic acid in postmenopausal osteoporosis [letter] N Engl J Med. 2007;357(7):712–713. [PubMed]
124. Burr DB, Miller L, Grynpas M, et al. Tissue mineralization is increased following 1-year treatment with high doses of bisphosphonates in dogs. Bone. 2003;33(6):960–969. [PubMed]
125. Chapurlat RD, Arlot M, Burt-Pichat B, et al. Microcrack frequency and bone remodeling in postmenopausal osteoporotic women on long-term bisphosphonates: a bone biopsy study. J Bone Miner Res. 2007;22(10):1502–1509. [PubMed]
126. Odvina CV, Zerwekh JE, Rao DS, Maalouf N, Gottschalk FA, Pak CY. Severely suppressed bone turnover: a potential complication of alendronate therapy. J Clin Endocrinol Metab. 2005 Mar;90(3):1294–1301. Epub 2004 Dec 14. [PubMed]
127. Armamento-Villareal R, Napoli N, Panwar V, Novack D. Suppressed bone turnover during alendronate therapy for high-turnover osteoporosis [letter] N Engl J Med. 2006;355(19):2048–2050. [PubMed]
128. Whitson HE, Lobaugh B, Lyles KW. Severe hypocalcemia following bisphosphonate treatment in a patient with Paget’s disease of bone. Bone. 2006 Oct;39(4):954–958. Epub 2006 Jun 12. [PubMed]
129. Jones SG, Dolan G, Lengyel K, Myers B. Severe increase in creatinine with hypocalcaemia in thalidomide-treated myeloma patients receiving zoledronic acid infusions [letter] Br J Haematol. 2002;119(2):576–577. [PubMed]
130. Mishra A, Wong L, Jonklaas J. Prolonged, symptomatic hypocalcemia with pamidronate administration and subclinical hypoparathyroidism. Endocrine. 2001;14(2):159–164. [PubMed]
131. Maalouf NM, Heller HJ, Odvina CV, Kim PJ, Sakhaee K. Bisphosphonate-induced hypocalcemia: report of 3 cases and review of literature. Endocr Pract. 2006;12(1):48–53. [PubMed]
132. Hewitt RE, Lissina A, Green AE, Slay ES, Price DA, Sewell AK. The bisphosphonate acute phase response: rapid and copious production of proinflammatory cytokines by peripheral blood gd T cells in response to aminobisphosphonates is inhibited by statins. Clin Exp Immunol. 2005;139(1):101–111. [PMC free article] [PubMed]
133. Barrera BA, Wilton L, Harris S, Shakir SA. Prescription-event monitoring study on 13,164 patients prescribed risedronate in primary care in England. Osteoporos Int. 2005 Dec;16(12):1989–1998. Epub 2005 Aug 31. [PubMed]
134. US Food and Drug Administration. Information on bisphosphonates (marketed as Actonel, Actonel+Ca, Aredia, Boniva, Didronel, Fosamax, Fosamax+D, Reclast, Skelid, and Zometa) Jan 72008. [Accessed July 18, 2008]. http://ovha.vermont.gov/for-providers/i3a_fda_gov_cder_drug_infopage_bisphosphonates_default.pdf.
135. Wysowski DK, Chang JT. Alendronate and risedronate: reports of severe bone, joint, and muscle pain [letter] Arch Intern Med. 2005;165(3):346–347. [PubMed]
136. Smetana S, Michlin A, Rosenman E, Biro A, Boaz M, Katzir Z. Pamidronate-induced nephrotoxic tubular necrosis—a case report. Clin Nephrol. 2004;61(1):63–67. [PubMed]
137. Chang JT, Green L, Beitz J. Renal failure with the use of zoledronic acid [letter] N Engl J Med. 2003;349(17):1676–1679. [PubMed]
138. Barone A, Giusti A, Pioli G, et al. Secondary hyperparathyroidism due to hypovitaminosis D affects bone mineral density response to alendronate in elderly women with osteoporosis: a randomized controlled trial. J Am Geriatr Soc. 2007;55(5):752–757. [PubMed]
139. Holick MF. Vitamin D deficiency. N Engl J Med. 2007;357(3):266–281. [PubMed]
140. National Institutes of Health. Optimal calcium intake. NIH Consens Statement. 1994. [Accessed July 18, 2008]. pp. 1–31. http://www.ncbi.nlm.nih.gov/books/bv.fcgi?rid=hstat4.chapter.13595. [PubMed]
141. National Osteoporosis Foundation’s. Updated Recommendations for Calcium and Vitamin D Intake. July 262007. [Accessed July 18, 2008]. http://216.247.61.108/prevention/calcium_and_VitaminD.htm.
142. Dietary Reference Intakes (DRIs): Summary Tables; 2004.

Additional Topics: Acute Back Pain
Back pain is one of the most prevalent causes of disability and missed days at work worldwide. Back pain is the second most common reason for doctor office visits, outnumbered only by upper-respiratory infections. Approximately 80 percent of the population will experience back pain at least once throughout their life. The spine is a complex structure made up of bones, joints, ligaments, and muscles, among other soft tissues. Because of this, injuries and/or aggravated conditions, such as herniated discs, can eventually lead to symptoms of back pain. Sports injuries or automobile accident injuries are often the most frequent cause of back pain, however, sometimes the simplest of movements can have painful results. Fortunately, alternative treatment options, such as chiropractic care, can help ease back pain through the use of spinal adjustments and manual manipulations, ultimately improving pain relief.

EXTRA IMPORTANT TOPIC: Hip Pain Chiropractic Treatment
Post Disclaimer
General Disclaimer, Licenses and Board Certifications *
Professional Scope of Practice *
The information herein on "Bisphosphonates: Mechanism of Action and Role in Clinical Practice" is not intended to replace a one-on-one relationship with a qualified health care professional or licensed physician and is not medical advice. We encourage you to make healthcare decisions based on your research and partnership with a qualified healthcare professional.
Blog Information & Scope Discussions
Welcome to El Paso's Premier Wellness and Injury Care Clinic & Wellness Blog, where Dr. Alex Jimenez, DC, FNP-C, a Multi-State board-certified Family Practice Nurse Practitioner (FNP-BC) and Chiropractor (DC), presents insights on how our multidisciplinary team is dedicated to holistic healing and personalized care. Our practice aligns with evidence-based treatment protocols inspired by integrative medicine principles, similar to those on this site and on our family practice-based chiromed.com site, focusing on naturally restoring health for patients of all ages.
Our areas of multidisciplinary practice include Wellness & Nutrition, Chronic Pain, Personal Injury, Auto Accident Care, Work Injuries, Back Injury, Low Back Pain, Neck Pain, Migraine Headaches, Sports Injuries, Severe Sciatica, Scoliosis, Complex Herniated Discs, Fibromyalgia, Chronic Pain, Complex Injuries, Stress Management, Functional Medicine Treatments, and in-scope care protocols.
Our information scope is multidisciplinary, focusing on musculoskeletal and physical medicine; wellness; contributing etiological viscerosomatic disturbances within clinical presentations; associated somato-visceral reflex clinical dynamics; subluxation complexes; sensitive health issues; and functional medicine articles, topics, and discussions.
We provide and present clinical collaboration with specialists from various disciplines. Each specialist is governed by their professional scope of practice and licensure jurisdiction. We use functional health & wellness protocols to treat and support care for musculoskeletal injuries or disorders.
Our videos, posts, topics, and insights address clinical matters and issues that directly or indirectly relate to our clinical scope of practice.
Our office has made a reasonable effort to provide supportive citations and has identified relevant research studies that support our posts. We provide copies of supporting research studies upon request to regulatory boards and the public.
We understand that we cover matters that require an additional explanation of how they may assist in a particular care plan or treatment protocol; therefore, to discuss the subject matter above further, please feel free to ask Dr. Alex Jimenez, DC, APRN, FNP-BC, or contact us at 915-850-0900.
We are here to help you and your family.
Blessings
Dr. Alex Jimenez DC, MSACP, APRN, FNP-BC*, CCST, IFMCP, CFMP, ATN
email: [email protected]
Multidisciplinary Licensing & Board Certifications:
Licensed as a Doctor of Chiropractic (DC) in Texas & New Mexico*
Texas DC License #: TX5807, Verified: TX5807
New Mexico DC License #: NM-DC2182, Verified: NM-DC2182
Multi-State Advanced Practice Registered Nurse (APRN*) in Texas & Multi-States
Multi-state Compact APRN License by Endorsement (42 States)
Texas APRN License #: 1191402, Verified: 1191402 *
Florida APRN License #: 11043890, Verified: APRN11043890 *
Colorado License #: C-APN.0105610-C-NP, Verified: C-APN.0105610-C-NP
New York License #: N25929, Verified N25929
License Verification Link: Nursys License Verifier
* Prescriptive Authority Authorized
ANCC FNP-BC: Board Certified Nurse Practitioner*
Compact Status: Multi-State License: Authorized to Practice in 40 States*
Graduate with Honors: ICHS: MSN-FNP (Family Nurse Practitioner Program)
Degree Granted. Master's in Family Practice MSN Diploma (Cum Laude)
Dr. Alex Jimenez, DC, APRN, FNP-BC*, CFMP, IFMCP, ATN, CCST
(Board Certified: Family Practice Nurse Practitioner—Multistate)*
(Licensed Nurse Practitioner & Chiropractor - Multistate)*
Clinical Director
Digital Business Card
Dr. Maria Cardenas, MD
(Board Certified: Internal Medicine)
(Licensed Medical Doctor)
Medical Director, Clinical Director & Collaborative Physician
NPI # 1164426749
MD License #: J2933
Licenses and Board Certifications:
MD: Medical Doctor
DC: Doctor of Chiropractic
APRNP: Advanced Practice Registered Nurse
FNP-BC: Family Practice Specialization (Multi-State Board Certified)
RN: Registered Nurse (Multi-State Compact License)
CFMP: Certified Functional Medicine Provider
MSN-FNP: Master of Science in Family Practice Medicine
MSACP: Master of Science in Advanced Clinical Practice
IFMCP: Institute of Functional Medicine
CCST: Certified Chiropractic Spinal Trauma
ATN: Advanced Translational Neutrogenomics
Memberships & Associations:
TCA: Texas Chiropractic Association: Member ID: 104311
AANP: American Association of Nurse Practitioners: Member ID: 2198960
ANA: American Nurse Association: Member ID: 06458222 (District TX01)
TNA: Texas Nurse Association: Member ID: 06458222
NPI: 1205907805
| Primary Taxonomy | Selected Taxonomy | State | License Number |
|---|---|---|---|
| No | 111N00000X - Chiropractor | NM | DC2182 |
| Yes | 111N00000X - Chiropractor | TX | DC5807 |
| Yes | 363LF0000X - Nurse Practitioner - Family | TX | 1191402 |
| Yes | 363LF0000X - Nurse Practitioner - Family | FL | 11043890 |
| Yes | 363LF0000X - Nurse Practitioner - Family | CO | C-APN.0105610-C-NP |
| Yes | 363LF0000X - Nurse Practitioner - Family | NY | N25929 |
Dr. Alex Jimenez, DC, APRN, FNP-BC*, CFMP, IFMCP, ATN, CCST
(Board Certified: Family Practice Nurse Practitioner—Multistate)*
(Licensed Nurse Practitioner & Chiropractor - Multistate)*
Clinical Director
Digital Business Card
Dr. Maria Cardenas, MD
(Board Certified: Internal Medicine)*
(Licensed Medical Doctor)*
Medical Director, Clinical Director & Collaborative Physician
NPI # 1164426749
MD License #: J2933
📆 Schedule Appointment: Schedule 24/7 (Click Here)